Introducing the OBS® Sterile Disposable Skin Stapler! This innovative tool is all about delivering top-notch precision, control, and efficiency, making it a go-to for surgeons and healthcare professionals in various surgical fields. It’s designed with reliability and user-friendliness in mind, simplifying the suturing process while ensuring the best possible outcomes for patients.
Key Features & Benefits:
Ergonomic Design: Its sleek, lightweight build allows for easy one-handed operation and precise control.
Precision & Safety: Enjoy smooth stapling with accurate needle withdrawal, helping to prevent any tissue damage.
Aesthetic Closure: It creates neat, uniform wound edges, leading to minimal scarring and better cosmetic results.
Superior Biocompatibility: Crafted from high-quality materials that reduce inflammation and ensure excellent compatibility with tissue.
Faster Recovery: The staples are quick and easy to remove, which promotes faster healing and less discomfort for patients.
Patient-Centered Comfort: It minimizes pain during closure while enhancing the overall efficiency of the surgical workflow.
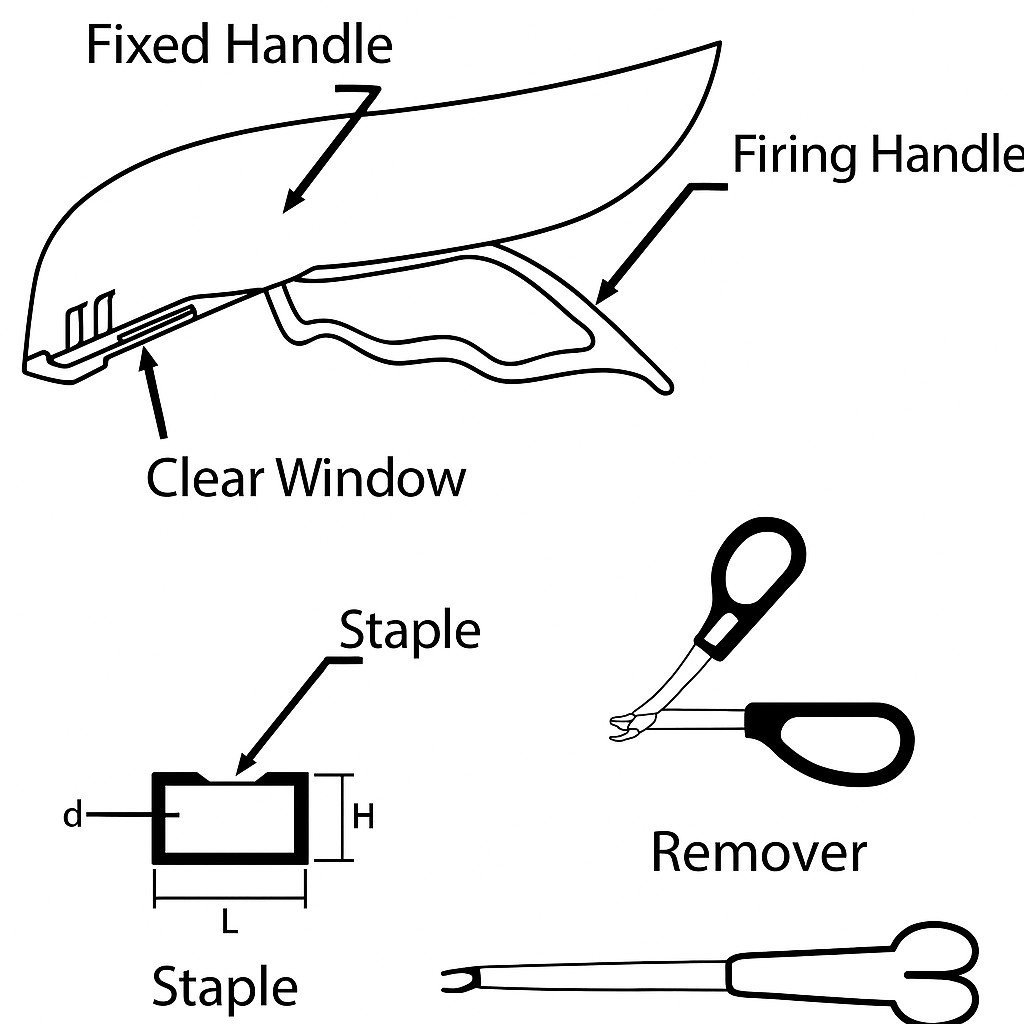
The Disposable Skin Stapler is crafted for the swift and effective closure of skin incisions or wounds. Made mainly from stainless steel staples and featuring an ergonomic handle, it allows for precise alignment of the skin edges during surgical procedures.
Warnings and Precautions
This product is sterile and meant for single use only. Please do not reuse, reprocess, or attempt to re-sterilize it under any circumstances. Doing so could compromise the device’s structural integrity, leading to possible malfunctions, patient injuries, infections, or even fatalities.
Reusing or re-sterilizing increases the risk of contamination and cross-infection, which could spread infectious diseases between patients.
The device is sterilized with ethylene oxide (EO) and has a shelf life of three years. Avoid using it if the expiration date has passed or if the sterile packaging is damaged, opened, or compromised.
Do not conduct MRI examinations on patients with staples in place, as the safety of the stainless steel staples in MRI conditions has not been confirmed.
Staple removal should only occur once the wound has fully healed and should be done solely at the discretion of the attending surgeon.
Instructions for Use — Suturing Procedure
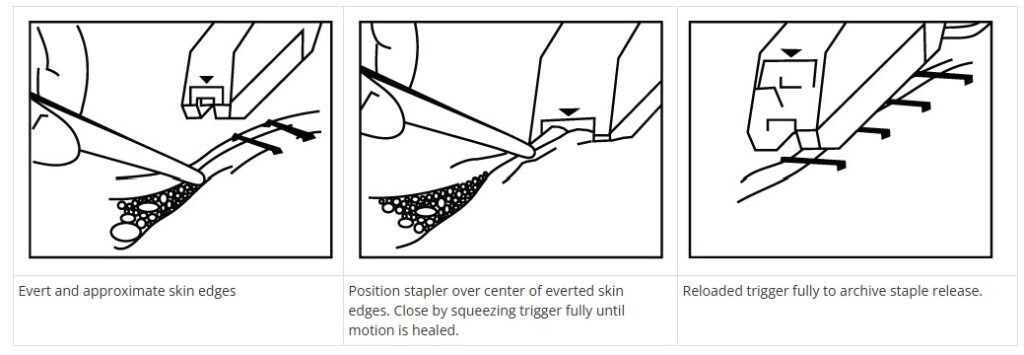
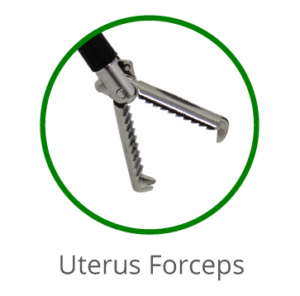
Gently bring the skin edges together using tissue forceps until they are slightly everted.
Position the stapler directly over the everted skin edges, making sure to align the device’s arrow with the center of the incision.
Squeeze the handle all the way until the firing mechanism activates and the handle is completely closed.
Release the handle and carefully lift the device away from the wound once the staple has been properly placed.
Staple Removal Procedure
Start by positioning the lower jaw of the staple remover right under the arch of the staple.
Next, squeeze the handle all the way until the legs of the staple bend outward and the handle is fully closed.
Make sure the staple is completely disengaged and reshaped before you gently lift it away from the skin.
Packaging and Storage
The instrument is sterilized with Ethylene Oxide (EO) and has a shelf life of three years from the manufacturing date, which you can find on the package.
Keep the product stored in a cool, dry, and well-ventilated space, away from direct sunlight and any corrosive gases.
To maintain the product’s integrity and sterility, ensure that the relative humidity in the storage area stays below 80%.
Staple Removal Procedure
Start by positioning the lower jaw of the staple remover right under the arch of the staple.
Next, squeeze the handle all the way until the legs of the staple bend outward and the handle is fully closed.
Make sure the staple is completely disengaged and reshaped before you gently lift it away from the skin.
Packaging and Storage
The instrument is sterilized with Ethylene Oxide (EO) and has a shelf life of three years from the manufacturing date, which you can find on the package.
Keep the product stored in a cool, dry, and well-ventilated space, away from direct sunlight and any corrosive gases.
To maintain the product’s integrity and sterility, ensure that the relative humidity in the storage area stays below 80%.







Reviews
There are no reviews yet.